What is TRAC?
In the fight against cancer, understanding how tumors respond to therapeutic strategies is of great importance. As patients undergo various cancer treatments, changes in tumor size and attributes can be documented and summarized in a report known as Tumor Response Assessment Criteria (TRAC).
TRAC reports are generated from 3D imaging measurements, representing the medical imaging perspective on understanding tumor response over time. These reports utilize a technique known as longitudinal analysis, which involves the collection of data points over time to illustrate the overall trajectory of tumor response. This approach enables healthcare providers attain a deeper insight into a patient’s cancer journey in response to treatment, thereby equipping them to make informed adjustments to treatment plans and predict patient outcomes with greater precision.
Learn more about longitudinal analysis here: https://3dqlab.stanford.edu/longitudinal-analysis/
Figure A (Right): A visual representation of longitudinal analysis, demonstrating the change of size in three different tumor segmentations over time.

How is TRAC performed?
When a physician requests a TRAC report, a technologist begins by establishing a baseline by measuring the dimensions of the target tumor(s) on the scan, as well as identifying any additional non-target tumors. Approximately three months into a patient’s cancer treatment, a follow-up scan is conducted, and these measurements are repeated. The collected data is analyzed using specific tumor response criteria tailored to the type of cancer and the administered treatment. The resulting findings are securely stored within our system and can be presented in a variety of formats, including tabulated summaries, stacked charts, and galleries of snapshots generated from DICOM images. Presented below are examples of TRAC report pages for reference:
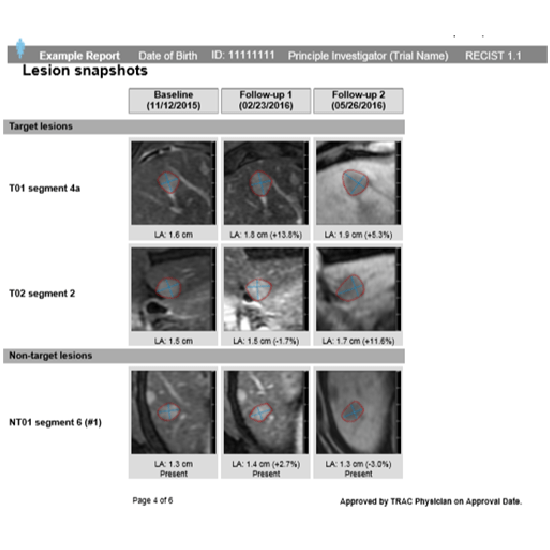

Figure B: Example TRAC report images of a target and non-target lesion’s baseline and follow-up measurements.
Figure C: Example TRAC report graph demonstrating the size of three target lesions from baseline through twelve follow-up measurements.


Figure D: Example TRAC report graph demonstrating a partial response trajectory.
Figure E: Example TRAC report table displaying all baseline and follow-up findings.
How are results defined?
Tumor response criteria are standardized guidelines used to assess how tumors react to cancer treatments, creating a consistent framework for healthcare professionals to monitor changes in tumor size, characteristics, and behavior resulting from treatments such as immunotherapy. Multiple response criteria have been developed as the complexity of cancer has become more understood, as the way tumors respond to treatment can vary due to several factors. These criteria have been specifically crafted to accommodate the distinct nature of various cancer types, diverse treatment approaches, and the continually evolving landscape of cancer therapies.
These criteria serve several roles:
- First and foremost, they aid in evaluating treatment effectiveness by helping healthcare providers understand changes in tumors:
- Tumors shrinking (partial response)
- Disappearing (complete response)
- Remaining stable (no significant change)
- Growing (progressive disease) in response to therapy
- Secondly, they enable informed treatment decisions, offering guidance to physicians on whether to:
- Continue with the current treatment
- Modify the treatment plan
- Explore alternative therapies
Outlined below are the various tumor response criteria employed by the 3DQ Lab.

Figure F (Above): Venn diagram of the tumor response criteria used by the 3DQ Lab.
Morphologic / Anatomic
Morphologic and anatomic criteria are used to categorize the physical features of a tumor, such as size and shape.
RECIST 1.1 (Response Evaluation Criteria in Solid Tumors 1.1):
RECIST 1.1 uses imaging measurements to assess structural changes in solid tumors.
mRECIST Mesothelioma (Modified RECIST for Mesothelioma):
mRECIST Mesothelioma is designed to assess the structure of specific cancers affecting the lining of the lungs, abdomen, or heart.
mRECIST HCC
mRECIST HCC is focused on hepatocellular carcinoma (liver cancer) and specific treatments like transarterial chemoembolization (TACE).
Immune-Related Response
Immune-related response criteria involves categorizing the specific patterns of tumor response and changes in the context of cancer treatment with immunotherapies, particularly immune checkpoint inhibitors. These treatments work by harnessing the patient’s immune system to target and attack cancer cells.
irRC (Immune-Related Response Criteria):
irRC is a set of criteria designed for assessing tumor response in patients undergoing immunotherapy, particularly immune checkpoint inhibitors. It considers unique response patterns seen with immunotherapies, including pseudoprogression.
imRECIST (Modified RECIST for Immunotherapies):
imRECIST is another modification of RECIST, tailored for assessing tumor response in patients receiving immunotherapies like immune checkpoint inhibitors. It accounts for immune-related response patterns.
iRECIST (Immune RECIST):
iRECIST is a refinement of RECIST criteria developed for assessing tumor response in immunotherapy trials. It addresses the challenges of immune-related responses, including the concept of unconfirmed responses.
irRECIST (Immune-Related RECIST):
irRECIST is an extension of RECIST designed for immunotherapy trials. It includes immune-specific criteria for assessing tumor response and progression and is primarily used in clinical trials.
Functional / Physiologic
Functional criteria assess the tumor or patient’s body functions in response to treatment, looking at factors like blood flow, organ function, or metabolic activity. Physiologic criteria consider the body’s physiological changes in response to treatment, evaluating overall health.
Cheson Criteria:
The Cheson criteria is used for evaluating treatment response in lymphomas, particularly non-Hodgkin lymphoma and Hodgkin lymphoma. It includes guidelines for assessing lymph node size, spleen size, and other factors specific to lymphomas.
Lugano Criteria:
The Lugano criteria is an update to the Cheson criteria and is used for evaluating treatment response in lymphomas, incorporating both anatomical and metabolic information. It defines response categories such as complete metabolic response (CMR) and partial metabolic response (PMR).
LYRIC (Lymphoma Response to Immunomodulatory Therapy Criteria) (Both Immune-Related and Functional / Physiological Responses):
The LYRIC criteria is used for assessing lymphoma response to immunomodulatory therapy, such as lenalidomide. It is specific to lymphoma treatment assessment.
Learn more about our TRAC program here: https://3dqlab.stanford.edu/trac/
3DQ Lab – Grant Building
3DQ Lab – Clark Building
Learn More About the Lab
Copyright © Stanford University



